
Regulatory affairs can be complicated, as many FDA guidelines are just that – guidelines. As site personnel, we frequently come across unique scenarios. It is often tough to figure out appropriate responses while striving to maintain regulatory compliance, especially when the guidelines are vague. Your site’s regulatory binder for a trial is always the first set of documents reviewed during routine audits and inspections, including CRA visits. It is essential to consistently maintain your trial master file.
We have outlined a few best practices on how to prepare your regulatory binder for your next study:
- Reverse Chronological Order: Store items in reverse order so that the most recent documents are located towards the front.
- Electronic Documents: If you are using electronic documents, it may help to insert a paper placeholder to indicate that the documents for that section have electronic copies.
- Create Tabs: Create tabs for each section of the binder as noted below. You may need to use multiple binders; just be sure to label each one with the protocol number, sponsor, and binder number in order to stay organized!
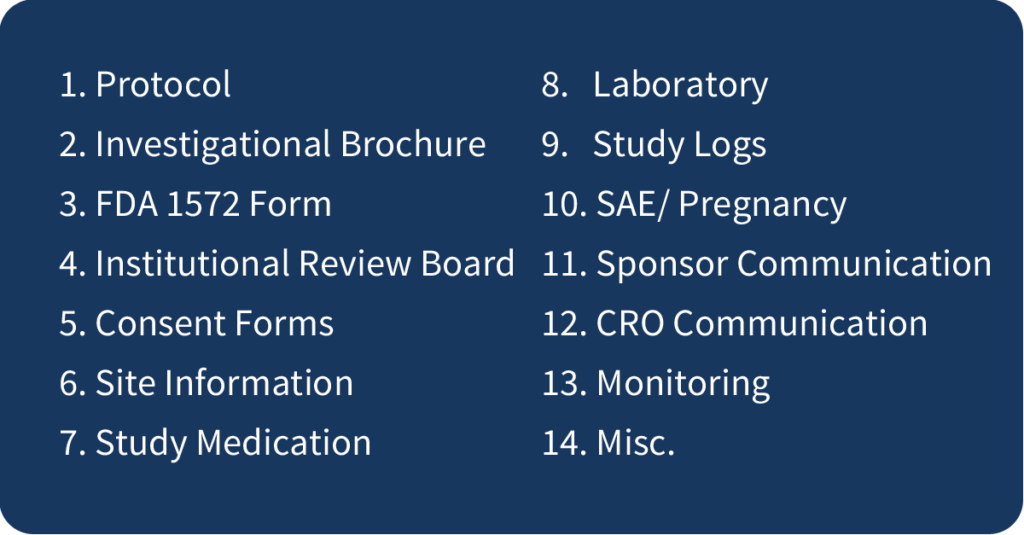
GOOD RULE OF THUMB: Just like your patients’ source charts, your regulatory binders are the storybook of the trial and the papers inside are the chapters. Every document in the files should flow cohesively. Like always, if it is not documented, it never happened.
In our recent webinar, Kasi Bettencourt, Project Director at BTC Network, addressed these issues along with other common regulatory questions. This scenario-based webinar covered what to do when presented with one of these “gray areas” and offered advice for how to address future situations as they arise.
We also reviewed other important aspects of regulatory affairs, including maintenance of a study regulatory binder and how to address protocol deviations. The study regulatory binder – along with the patient source – is the “storybook” of the trial and it is imperative that sites organize these the correct way and do not leave anything out. It is also essential that protocol deviations, minor or major, are logged appropriately. We discussed everything from how to classify deviations to when it is required to report to the IRB.
To learn more about best practices for regulatory affairs, don’t forget to watch our webinar, Regulatory Affairs: The Big Picture, on demand by clicking on the button below!
About BTC Network
BTC Network is North America’s leading clinical research group. Based just outside of Boston, Massachusetts, our network includes more than 50 multi-therapeutic sites. All BTC research sites collaborate with or are part of private practices, giving us access to over 100,000 patients.



